Chronic lymphocytic leukaemia (CLL) and small lymphocytic lymphoma (SLL)
This information is about chronic lymphocytic leukaemia (CLL) and small lymphocytic lymphoma (SLL), which are different forms of the same illness. They are often grouped together as a type of low-grade non-Hodgkin lymphoma.
On this page
What is CLL/SLL?
Chronic lymphocytic leukaemia (CLL) and small lymphocytic lymphoma (SLL) are slow-growing types of blood cancer. They develop when white blood cells called lymphocytes grow out of control. Lymphocytes are part of your immune system. They travel around your body in your lymphatic system, helping you fight infections. There are two types of lymphocyte:
T lymphocytes (T cells) and B lymphocytes (B cells).
CLL and SLL are different forms of the same illness. They develop when B cells that don’t work properly build up in different parts of your body.
- In CLL, the abnormal B cells build up in your blood and bone marrow. This is why it’s called ‘leukaemia’ – after ‘leucocytes’: the medical name for white blood cells.
- In SLL, the abnormal B cells mainly build up in your lymph nodes. This is why it’s called ‘lymphoma’.
CLL and SLL are slow-growing (low-grade or ‘indolent’) cancers. Many doctors group them together as a type of non-Hodgkin lymphoma. They usually behave like a long-term (chronic) condition that needs treatment from time-to-time to keep it under control.
CLL and SLL are treated in the same way. In this information, we use the term ‘CLL/SLL’ to refer to both, except where there are important differences.
Hear Professor George Follows, Consultant Haematologist at Cambridge University Hospital, talk about CLL and SLL
Who gets CLL/SLL?
Around 4,500 people are diagnosed with CLL/SLL every year in the UK – more than 12 people each day. It affects nearly twice as many men as women. It is more common in people over 60, with an average age at diagnosis of 72 years. CLL/SLL is rare in young people.
In most cases, there is no known cause of CLL/SLL. People who have a close relative (parent, brother, sister or child) with CLL/SLL have a higher chance of developing it themselves, but the risk is still very low.
Some people have a low level of abnormal B cells in their blood, a condition called monoclonal B-lymphocytosis (MBL). There are no symptoms for MBL and so most people with this condition never know they have it. Each year, around 2 in 100 people with MBL go on to develop CLL/SLL. This is more likely to happen in people with a higher number of these abnormal B cells (sometimes called ‘high-count MBL’) so people with MBL have regular blood tests to check their level of abnormal cells.
Symptoms of CLL/SLL
Most people have no symptoms when they are diagnosed with CLL/SLL. It’s usually found by chance when doctors are doing a test for another reason.
As abnormal B cells build up, you might start to develop symptoms, although some people never do. Any symptoms tend to be mild at first and gradually get worse over a period of months or years. You might feel generally unwell, or notice symptoms like:
- fatigue (extreme tiredness)
- weight loss
- fevers
- night sweats
- frequent infections.
Other symptoms depend on where in your body the abnormal cells build up.
- In your lymph nodes – causing them to swell. You might feel these as lumps. They can develop anywhere in your body but are most often found in your neck, armpit or groin.
- In your spleen (an organ in your lymphatic system) – you might have a change in appetite and feel pain or a lump at the top-left of your tummy (abdomen).
- In your bone marrow – they can take up the space needed for healthy blood cells to develop, this means your body might not be able to make enough blood cells and you might develop low blood counts:
- anaemia (low red blood cells), which can make you feel tired, breathless or dizzy
- thrombocytopenia (low platelets), which makes you more likely to bruise and bleed
- neutropenia (low neutrophils – a type of white blood cell), which might make you pick up infections more easily than usual, and can make it harder to get rid of them.
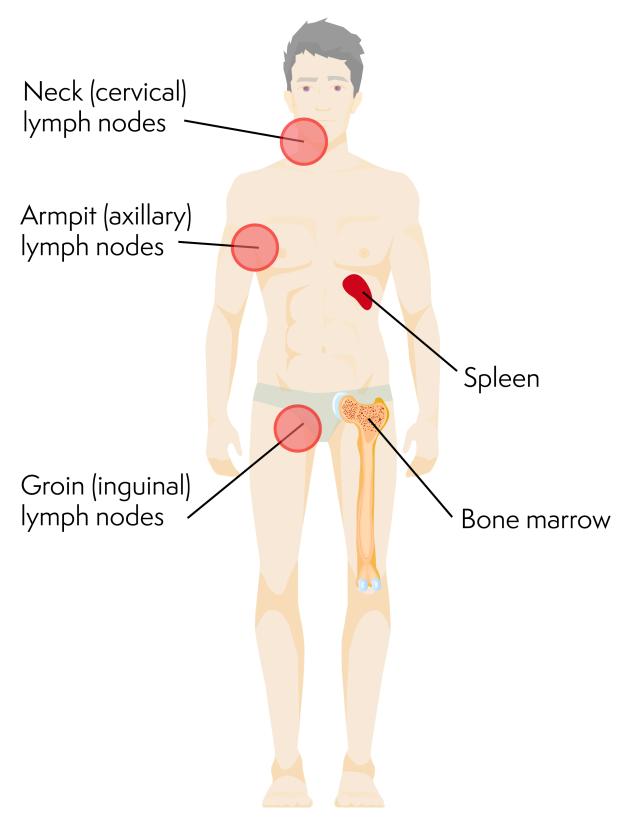
Parts of the body where CLL/SLL is often found
Some people with CLL/SLL develop low blood counts due to an autoimmune reaction. This is when your body makes antibodies that attack your own healthy cells. If the antibodies stick to blood cells, they can cause:
- low red blood cells (autoimmune haemolytic anaemia, or AIHA)
- low platelets (immune thrombocytopenic purpura, or ITP).
Low blood counts due to autoimmune conditions need different treatment from low blood counts caused by too many abnormal cells in your bone marrow.
Diagnosis and staging
You might be diagnosed with CLL/SLL by chance when you are having tests for something else, or your doctor may suspect CLL/SLL based on your symptoms. A range of tests can be used to confirm the diagnosis, understand the stage of your CLL/SLL and determine the treatment you are most likely to benefit from. It can be hard to distinguish between CLL and SLL and sometimes the diagnosis can change. As the treatment for CLL and SLL is the same, this is not something to be concerned about.
You usually have your tests done as an outpatient. It takes a few weeks to get all the results. Waiting for test results can be a worrying time, but it is important for your medical team to have all the information they need so they can plan the most appropriate treatment for you.
Blood tests
CLL can be diagnosed from a blood test where a specialist examines a sample of your blood under a microscope. SLL cannot be diagnosed using a blood test alone so other diagnostic tests will also be used, typically a lymph node biopsy.
You also have blood tests to look at your general health, measure your antibody levels, and find out how well your kidneys and liver are working. If you are starting treatment, you might have blood tests to check if you have any viral infections that could flare up during treatment.
Biopsies
A biopsy is a minor procedure to remove a sample of cells from your body. A doctor either uses a hollow needle to remove a ‘core’ of tissue from a lymph node (a ‘needle core biopsy’), or removes the whole node. Both types of biopsy are done under anaesthetic so you won’t feel any pain. This is usually a local anaesthetic, but occasionally you might have a general anaesthetic depending on where in the body the biopsy is taking place.
SLL is usually diagnosed by taking a sample of cells from a swollen lymph node, this is called a lymph node biopsy.
If you have low blood counts, you might have a bone marrow biopsy to check for lymphoma cells in your bone marrow. You might also have a bone marrow biopsy when you finish treatment, to check how well your lymphoma has responded. However, this isn’t always needed.
Specialist tests
Your blood and biopsy samples are examined by an expert CLL/SLL or lymphoma pathologist. The pathologist looks at the samples under a microscope and tests the abnormal B cells to find out if they have certain genetic changes. Some genetic changes mean CLL/SLL is likely to grow faster and need a different type of treatment. These are called ‘high-risk’ genetic changes.
Your medical team uses this information from your blood tests and biopsy to help choose the most appropriate treatment for you.
Other tests
Ultrasound or CT scans help your medical team find out which parts of your body are affected by CLL/SLL and how enlarged your lymph nodes, liver and spleen are. These scans can be used as a reference or ‘baseline’. You usually have another scan after any treatment to check how well you have responded to it.
Immunoglobulin (antibody) level tests can help your medical team check how well you are able to fight infections. Antibody levels can also indicate how your body may respond to treatment.
Staging of CLL/SLL
Your medical team uses the results of your tests and scans to work out how widespread your CLL/SLL is and how it is affecting you. This is called staging. Staging is important because it helps your medical team plan the best treatment for you.
Staging of SLL is the same as for other types of non-Hodgkin lymphoma. CLL is staged differently because it generally affects your blood rather than your lymph nodes. The staging system commonly used for CLL in the UK is called the Binet system. This is a three-step staging system based on the number of swollen lymph nodes you have and your blood test results. (A system called the Rai staging system is often used in the United States – you might see reference to this in information that is not from the UK).
The Binet system
- Stage A: You have fewer than three areas of ‘lymphoid swelling’ (swollen liver or spleen, or swollen lymph nodes in your neck, armpits or groin).
- Stage B: You have three or more areas of lymphoid swelling.
- Stage C: You have a low red blood cell count (anaemia), a low platelet count (thrombocytopenia) or both.
Outlook
CLL/SLL usually grows very slowly and there are lots of effective treatment options. Treatment generally aims to keep the lymphoma under control rather than curing it.
Most people live with CLL/SLL for many years, with some periods when they need treatment, and other periods when they do not. It is hard to predict how long it might be before you start treatment.

- Around 1 in 3 people need treatment soon after their diagnosis.
- Around 1 in 3 people need treatment at some point in the future.
- Around 1 in 3 people never need treatment.
In general, people with CLL/SLL at an earlier stage have a better outlook than those with more advanced CLL/SLL. Some people have particular genetic changes in their lymphoma cells that mean it grows faster and is likely to need more frequent treatment. Your doctor is best placed to advise you on your outlook based on your individual circumstances and test results.
Rarely, CLL/SLL transforms (changes) into a faster-growing type of lymphoma. This is known as ‘Richter transformation’. It happens to about 1 in every 100 people with CLL/SLL each year. Most of these cases transform to a type of diffuse large B-cell lymphoma (DLBCL) but they can occasionally transform to other non-Hodgkin lymphomas or Hodgkin lymphoma. Transformed CLL/SLL is more difficult to treat.
Treatment
CLL/SLL is slow-growing and there is rarely an urgent need for treatment. Some people might not need treatment for many years. Your medical team will consider carefully whether you need treatment straight away and what treatment is best for you.
Active monitoring (watch and wait)
Most people don’t have any symptoms when they are first diagnosed with CLL/SLL. If the CLL/SLL is not causing problems, you do not need treatment straight away. Instead, your medical team monitors you regularly until you need treatment. This is called active monitoring or watch and wait.
I was on active monitoring from August 2011 until 2015 when it was agreed that it was time to consider treatment.
Read more about Carl’s story.
Research shows that there is no benefit to starting treatment before you need it. All treatment also carries a risk of causing side effects, so doctors often advise delaying treatment for as long as possible.
If you are worried about your health at any time, contact your GP or medical team. You don’t have to wait for your next appointment.
You are likely to start treatment if:
- your symptoms get worse or become difficult to cope with
- your lymph nodes, spleen or liver become swollen enough to cause you symptoms
- you develop low blood counts
- the number of abnormal lymphocytes in your blood rises very quickly
- you have autoimmune anaemia or thrombocytopenia that is not responding to treatment.
When you finish your treatment, you go back onto active monitoring.
Treatment options
The treatment your medical team recommends depends on:
- the stage of your CLL/SLL
- how it is affecting you
- the particular genetic changes in your CLL/SLL cells
- any other illnesses you have
- the potential side effects or late effects (health problems that develop months or years after treatment) of the treatment
- your preferences.
Depending on your individual circumstances, you might need several different treatments during your illness. Treatment aims to reduce your symptoms and keep the CLL/SLL under control for as long as possible with as few side effects as possible. Most people have long periods of feeling well between courses of treatment.
Most people with CLL/SLL are treated with targeted drugs. Targeted treatments are medicines that have been specially designed to attack particular proteins on the surface or within the CLL/SLL cells. The proteins they attack are over-active in the CLL/SLL cells compared with healthy cells. These proteins are often important in helping the cancer cells grow and survive, so blocking them with the targeted drugs helps to kill the CLL/SLL cells specifically.
Targeted drugs used to treat CLL/SLL include:
- acalabrutinib (a BTK inhibitor), used on its own or in combination with obinutuzumab (an antibody therapy)
- zanubrutinib (a BTK inhibitor) used on its own
- ibrutinib (a BTK inhibitor), used on its own or in combination with rituximab (an antibody therapy), obinutuzumab (an antibody therapy) or venetoclax (another targeted drug)
- venetoclax (a BCL-2 inhibitor) used in combination with obinutuzumab (an antibody therapy).
These targeted therapies are usually taken as a tablet or capsule orally. Antibody therapies are usually given as an infusion into the vein.
When used alone you usually carry on taking the targeted drug unless you develop troublesome side-effects or until your CLL/SLL stops responding – they are therefore often known as continuous treatments. If the targeted drug is taken in combination with another treatment (an antibody therapy or another targeted drug), you usually take it for a set duration, often 1 to 2 years.
Other treatment options
A chemo-immunotherapy regimen known as FCR (fludarabine, cyclophosphamide and rituximab) may be an appropriate treatment option if you are fit enough and depending on your specific diagnosis.
Your medical team might ask you if you’d like to take part in a clinical trial. To find out more about clinical trials or to search for a trial that might be suitable for you, visit Lymphoma TrialsLink.
I was treated as part of a clinical trial. The researchers won’t know if it was better than standard treatment for many years but I felt I was doing my bit by taking part.
Hear Michael's experience of taking part in a clinical trial for SLL
Radiotherapy is rarely used for CLL but is sometimes used to treat SLL that is only affecting one part of your body. In this case, it can sometimes cure the lymphoma.
Treatment or prevention of symptoms
As well as treatments to control your lymphoma, you might have treatments to help relieve your symptoms or prevent infections. These might include:
- antibiotics, antifungal and antiviral drugs to prevent or treat infections
- vaccinations against flu and pneumonia
- growth factor (G-CSF) injections to boost your white blood cell count
- immunoglobulin replacement therapy to strengthen your immune system if your level of natural antibodies is low
- red blood cell transfusions or platelet transfusions to treat low blood counts
- treatments for autoimmune haemolytic anaemia and immune thrombocytopenic purpura.
Contact your medical team straightaway if you have any symptoms of infection. It is important that you get prompt treatment.
Follow-up
If your treatment is for a fixed duration (such a chemo-immunotherapy), you are likely to go back onto active monitoring if your CLL/SLL is under control.
If you are taking one of the continuous targeted treatments, you might stay on this treatment for a number of years. You have regular clinic appointments and blood tests to make sure your CLL/SLL is under control.
As part of your follow-up, your medical team check your general health and look for any signs that your CLL/SLL might be becoming more active again. This usually happens quite slowly. Your medical team check for changes in your blood test results, and whether you develop any new symptoms or new swollen lymph nodes.
Rarely, CLL/SLL can change to a more aggressive condition. If your doctor thinks this might have happened, they might arrange for you to have a PET/CT scan and another biopsy.
Relapsed or refractory CLL/SLL
Treatment for CLL/SLL is often effective. However, it is common for CLL/SLL to come back (relapse) and need more treatment. Occasionally, CLL/SLL doesn’t respond well to your first treatment. This is called ‘refractory’ CLL/SLL and is usually treated the same way as relapsed CLL/SLL. Some people with CLL/SLL need several courses of treatment during their illness.
The length of remission after successful treatment can vary a lot, so it can be difficult to predict how long it might be before you need more treatment. Some people stay in remission for several years but others need more treatment sooner. As new and more effective treatments for CLL/SLL become available, remissions are generally getting longer.
Treatment for relapsed or refractory CLL/SLL
When you need more treatment, your doctor considers the same factors as for your first treatment but also takes into account:
- what treatment you had before
- how well you responded to your previous treatment
- how your previous treatment affected you.
There are lots of different treatments available for CLL/SLL. If your CLL/SLL relapses, your medical team are likely to recommend a targeted treatment, different to the treatment option you had before. Options include:
- acalabrutinib (a BTK inhibitor)
- zanubrutinib (a BTK inhibitor)
- ibrutinib (a BTK inhibitor) on its own, or in combination with bendamustine (a chemotherapy) and rituximab (an antibody therapy)
- venetoclax (a BCL-2 inhibitor) in combination with rituximab (an antibody therapy), or alone if a BTK inhibitor is not suitable.
Your medical team might ask you if you’d like to take part in a clinical trial.
Rarely, your medical team might suggest an allogeneic stem cell transplant. This is an intensive form of treatment and is only suitable for people who are fit enough.
Treatment for transformed CLL/SLL
Transformed CLL/SLL can be difficult to treat. It does not usually respond well to treatment. Many people with transformed CLL/SLL are older and have had several previous courses of treatment so might not be fit enough for intensive treatment. The most likely treatment is chemotherapy, possibly followed by a donor (allogeneic) stem cell transplant if you are fit enough. If you are not fit enough for chemotherapy, you might be able to enter a clinical trial or you might consider palliative care, where you have treatment to control your symptoms.
Research
Scientists are testing many different targeted treatments in clinical trials for CLL/SLL, including some treatments that are already approved for other types of lymphoma. New types of treatment that are being tested in people with CLL/SLL include:
- B-cell receptor pathway inhibitors, which block signals that B cells send to help them divide or stay alive
- antibody therapies, including those that bind to two different targets (one on lymphoma cells and one on T cells, which helps the T cells find and destroy the lymphoma cells)
- new combinations of treatments
- CAR T-cell therapy, which involves genetically modifying your own T cells so they can recognise and kill lymphoma cells.
Some of these might be available to you through a clinical trial. Speak to your doctor if you are interested in taking part in a clinical trial. To find out more about clinical trials or to search for a trial that might be suitable for you, visit Lymphoma TrialsLink.
