CAR-T cell therapy
This information is about CAR-T cell therapy, a personalised treatment used for some types of lymphoma that have come back or not responded after previous treatment.
On this page
What is CAR-T cell therapy?
CAR-T cell therapy is a type of treatment that uses cells from your own immune system to fight lymphoma. They are called T cells, or T lymphocytes.
T cells are a type of white blood cell. As well as killing germs, T cells usually recognise and kill any of your own cells that have become abnormal (such as cancer cells). However, cancer cells are good at tricking your T cells, either by looking very similar to healthy cells or by sending signals that tell your T cells not to attack them.
In CAR-T cell therapy, your own T cells are collected and sent off to a lab. In the lab, they are genetically modified (changed) so they can recognise and stick to a particular protein on the surface of your lymphoma cells. These genetically modified T cells are known as ‘CAR T cells’. CAR stands for ‘chimeric antigen receptor’.
After they have been modified, the CAR T cells are grown in the lab until there are enough of them to treat your lymphoma. They are then given back to you, like a blood transfusion. When they stick to the lymphoma cells in your body, the CAR T cells are activated and kill the lymphoma cells.
Who might have CAR-T cell therapy?
CAR-T cell therapy is used to treat some types of lymphoma that have come back (relapsed) or not responded (refractory). It is a very intensive type of treatment and you have to be fit enough to have it.
You might be eligible for CAR-T cell therapy if you have:
- relapsed or refractory diffuse large B-cell lymphoma (DLBCL)
- relapsed or refractory primary mediastinal large B-cell lymphoma (PMBL) after at least two previous courses of treatment
- relapsed or refractory mantle cell lymphoma (to be eligible for CAR-T cell therapy, your previous treatment must have included a type of targeted drug called a BTK-inhibitor, such as ibrutinib).
The exact CAR-T cell therapy you might have depends on the type of lymphoma you have. Three different CAR-T cell therapies are currently approved in the UK:
- tisagenlecleucel (also known as tis-a-cel or Kymriah®)
- axicabtagene ciloleucel (also known as axi-cel or Yescarta®)
- brexucabtagene autoleucel (also known as KTE-X19 or Tecartus®).
CAR-T cell therapies are also being tested in clinical trials in other types of lymphoma. Visit Lymphoma TrialsLink to find out more about clinical trials and to search for a trial that might be suitable for you.
Having CAR-T cell therapy
CAR-T cell therapy can be an effective treatment option for some people. However, it is a very intensive type of treatment that can have serious, and occasionally life-threatening, side effects. You have to be fit enough to tolerate the treatment.
My consultant spoke to me about the option of CAR-T cell therapy. I wouldn’t be able to have this new treatment at my usual hospital, but would need to be transferred to a larger centre where this could be given. She explained that I would be a likely candidate for this therapy, but I would need to be assessed and ‘put forward’ for consideration.
If your doctor thinks you might benefit from CAR-T cell therapy, they refer you to a national panel of clinical experts. You will have tests to make sure you are well enough to have the treatment. These include blood tests to find out how well your liver and kidneys are working and to check if you have any viruses that might flare up during the treatment. You might have scans and other tests to check how well your heart and lungs are working. You might need a further biopsy.
Anne Hook from Lymphoma Action talks to Fiona, who had CAR-T cell therapy for DLBCL in 2019, about the selection process
This video is a collaboration between Gilead Sciences and Lymphoma Action. ©2022 Gilead Sciences Ltd. All rights reserved.
The panel also considers your general health, or ‘performance status’. This is often measured on a score of 0 to 4:
- 0: you are fully active and you can do all the things you used to do before you had lymphoma
- 1: you can't do heavy physical work, but you are mobile and you can do work that isn’t too physical
- 2: you can’t work but you can look after yourself and you’re up and about more than half the day
- 3: you need help to look after yourself and you spend more than half the day in bed or in a chair
- 4: you can’t look after yourself at all and you spend all your time in bed or in a chair.
The expert panel looks at all your information, including the results of your tests and scans, to help them decide if CAR-T cell therapy is suitable for you. To be eligible for CAR-T cell therapy, you must have a performance status score of 0 or 1 when you are first assessed.
CAR-T cell therapy can only be given at approved treatment centres that have the facilities and staff to administer it safely. If you are eligible for CAR-T cell therapy, you might have to travel some distance to have it.
CAR-T cell therapy involves a number of different steps:
Anne Hook from Lymphoma Action talks to Fiona about her experience of CAR-T cell therapy for DLBCL
This video is a collaboration between Gilead Sciences and Lymphoma Action. ©2022 Gilead Sciences Ltd. All rights reserved.
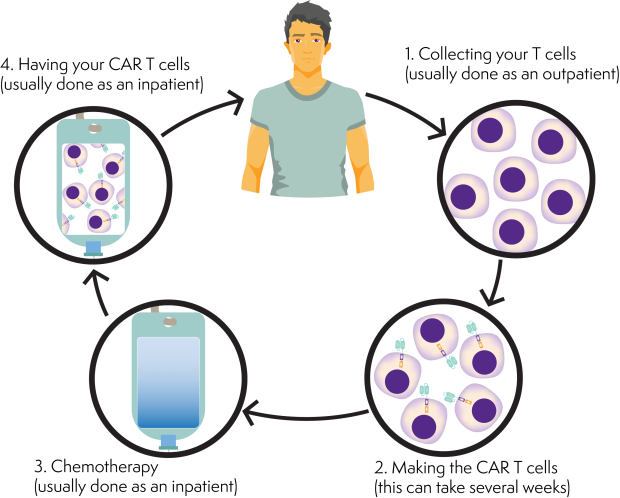
Step 1: Collecting your T cells
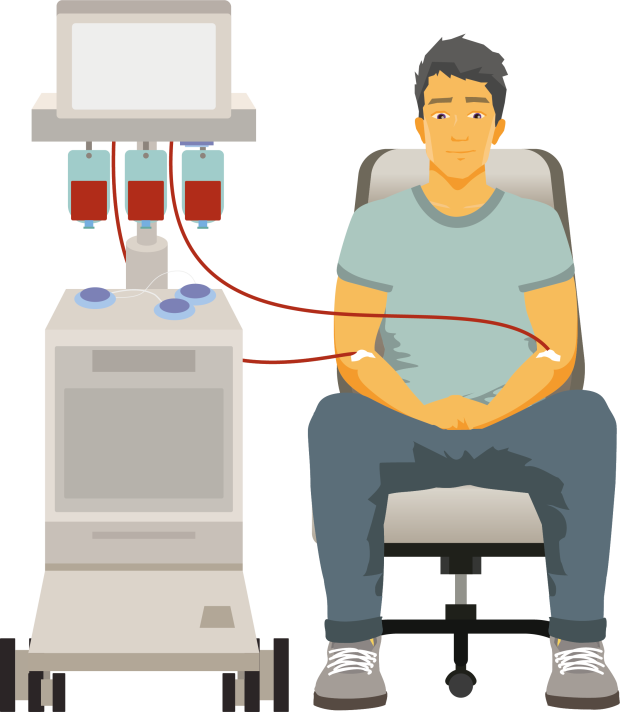
The first step is to collect your T cells. This process is called ‘apheresis’. You have it done as an outpatient and you can go home afterwards, unless you are already staying in hospital.
Depending on what medicines you are on, you might need to stop taking them a few days before your T cells are collected, to give the drugs time to leave your system. Often chemotherapy and steroids need to be avoided for a week or two before apheresis, depending on which CAR T product you are to have.
When you are ready to have your T cells collected, a doctor or nurse puts thin, flexible, plastic tubes into a vein in each of your arms (or they might use your central line, if you have one fitted). They connect these tubes to a cell separator machine. Your blood is taken from a tube in one arm and passes through the machine. The machine collects your T cells and returns the rest of your blood into your other arm. This process takes a few hours. You have to keep your arm still during this process and cannot move around much.
My T cells were taken from a line inserted into my groin at the apheresis centre early that morning and collected throughout the day – I was then able to go home.
Sometimes, it isn’t possible to collect enough T cells for CAR-T cell therapy. If this happens, your doctor will discuss your options with you.
Step 2: Making the CAR T cells
CAR-T cell treatments have to be made individually for each person. Your T cells are sent away to a lab for this. In the lab, your T cells are genetically modified to recognise a protein on your lymphoma cells. They are then grown until there are enough of them to treat your lymphoma, and frozen.
Making and growing the CAR T cells can take several weeks. During this time, you might need chemotherapy or radiotherapy to keep your lymphoma under control. This is called ‘bridging therapy’.
Sometimes, the lab isn’t able to successfully make the CAR T cells. If this happens, your doctor will discuss your options with you.
Step 3: Chemotherapy
Once your CAR T cells are ready, the lab send them back to the hospital where you’re being treated. The cells are frozen and can be stored for several months.
When they arrive, your doctor checks that you are still well enough to have them. You must have a performance status score of 0, 1 or 2 at this point. If you are not well enough to have the CAR T cells, your medical team might need to delay your treatment or discuss other treatment options with you.
If you are still well enough, the next step is to have chemotherapy to reduce the number of white blood cells in your body. This prepares your body for the CAR T cells so they can multiply more efficiently and work more effectively. It is called ‘lymphodepleting chemotherapy’.
The most common chemotherapy is a combination of fludarabine and cyclophosphamide, but other chemotherapy drugs are sometimes used. You usually have it through a drip into a vein, or through a central line if you have one. You have it as an outpatient over 3 to 5 days.
Step 4: Having your CAR T cells
Once you’ve finished your lymphodepleting chemotherapy, you are ready to have your CAR T cells.
- You have pre-medication first to help prevent any reactions. This is usually paracetamol and antihistamines (anti-allergy medicines).
- Then you have a single dose of CAR T cells through a drip into a vein, or through a central line if you have one. It only takes a few minutes.
The CAR-T cells were delivered back to me after 5 weeks, having been genetically modified on the other side of the Atlantic! It was dramatic! They are brought in in what looks like a milk churn! The bag of cells is placed in a water bath at body temperature. Once at this temperature they are delivered via the central line and a drip. The whole procedure took around 8 minutes from thawing to completion of delivery.
You are monitored carefully in hospital during and after the treatment. Most people stay in hospital for at least 10 days. After you leave hospital, you need to stay close by (usually within an hour’s travel) for at least 4 weeks after having your CAR T cells, in case you develop side effects. If you live more than an hour away, you might need to arrange accommodation closer to the hospital for a few weeks. Your hospital team might be able to help you arrange this.
You will also need to have someone with you at all times until 30 days after your CAR T infusion because of the possibility of delayed side effects.
Your medical team will tell you what to look out for and who to contact if you have any concerns.
Possible side effects
CAR-T cell therapy can cause serious side effects. Around 1 in 5 people who have CAR-T cell therapy need treatment on an intensive care unit (ICU).
CAR-T cell therapy is only given in hospitals with the facilities and staff to treat side effects effectively. You are monitored closely in hospital with regular blood tests for at least 10 days after having CAR-T cell treatment. You stay close to the hospital for another 2 weeks so you can be treated quickly if you develop any side effects.
The most common side effects of CAR T cells include:
This is not a complete list of side effects. Your medical team should tell you about the side effects you might experience and how to manage them. Ask as many questions as you want to make sure you understand everything.
Cytokine release syndrome
One of the most common complications of CAR-T cell therapy is called ‘cytokine release syndrome’ (CRS).
CRS happens when CAR T cells are activated and release chemicals called cytokines. These cytokines activate other immune cells, which also release cytokines. When lots of cytokines are released at once, they can overwhelm your body.
Signs and symptoms of CRS include:
- fever and chills
- rapid heart rate
- low blood pressure
- low oxygen levels in your body
- headache
- feeling or being sick.
Almost everyone treated with CAR-T cell therapy experiences some level of CRS. It most commonly develops within 10 days of having your CAR T cells. Most cases are mild and easily treated. However, severe or life-threatening reactions can occur. If you have a severe reaction, you might be transferred to the intensive care unit for treatments to help with your symptoms, such as oxygen and fluids. You might also have steroids and a drug called tocilizumab, which helps dampen down the immune response.
Around the sixth day I developed a high temperature, I felt unwell and slowly my energies seemed to drain away day-by-day. This response is expected and is ascribed to an effect called cytokine release syndrome (CRS). It can be very severe, requiring admission to the intensive care unit. Fortunately I avoided this! However I did feel unwell, at times, very unwell.
CRS usually gets better within a few days with treatment.
Nervous system problems
When lots of CAR T cells are activated, they release chemicals that can affect your nervous system. This is called ICANS (immune effector cell-associated neurotoxicity syndrome). Symptoms of ICANS include:
- confusion
- speech problems
- difficulty writing
- headaches and dizziness
- shaking or tremor
- movement difficulty.
Around 1 in 4 people treated with CAR-T cell therapy experience ICANS. It generally develops within 28 days of having your CAR T cells, often within just a few days. Symptoms are usually mild and get better on their own in a week or two.
Occasionally, people have more serious symptoms such as seizures (fits) or life-threatening problems such as swelling of the brain. Your medical team will monitor you closely for serious symptoms so they can start treatment promptly if you need it.
Daily questions are asked as a routine after CAR-T cell therapy to check for neurological complications, such as where one is, what day of the week it is, being asked to point to various things in the room, counting backwards from 100 in tens, even in sevens, plus writing a short sentence to check one’s neurological responses. My hand tended to shake as days went by, I noticed how my fine motor control had diminished.
If you have serious symptoms, you might need steroids and be admitted to the intensive care unit. Most people improve within a few days of treatment.
Low blood counts
A combination of chemotherapy and CAR-T cell therapy often leads to low blood counts. This affects your red blood cell count, your white blood cell count and your platelet count.
Most people have low blood counts during the first month after CAR-T cell therapy. Around 1 in 4 people have low blood counts that last several months. You will have blood tests to check your blood counts after you leave hospital. If your blood counts are very low or are causing problems, you might need blood transfusions or growth factors to help boost them.
Infections
After CAR-T cell therapy, your levels of a type of white blood cell called a B cell are likely to be low for a long time – sometimes several years. B cells make antibodies. Having a low B cell count and low antibody levels increases your risk of developing an infection.
While you are in hospital, your medical team monitor you closely for signs of infection. When you go home, they will tell you what signs to look out for and who to contact if you are worried you might have an infection. You will need antibiotics if you develop infection.
Contact your medical team straightaway if you have any signs of infection. It is important to get treatment promptly.
You will be given antibiotics and antiviral medicines to prevent infections (‘prophylactic’ treatment). If you have very low antibody levels, you might need treatment with donor antibodies (immunoglobulin replacement therapy).
After having CAR-T cell therapy, you might lose your immunity to illnesses you have been vaccinated against in the past. Your medical team might recommend that you repeat these vaccinations. They will tell you what they recommend depending on your individual circumstances.
Follow-up
Anne Hook from Lymphoma Action talks to Fiona about going home after having CAR-T cell therapy in 2019
This video is a collaboration between Gilead Sciences and Lymphoma Action. ©2022 Gilead Sciences Ltd. All rights reserved.
Most people who have CAR-T cell therapy stay in hospital for at least 10 days. After you leave, you must stay close to the hospital – within an hour’s travel – for 4 weeks after having your CAR T cells. You should not stay on your own during this time. Your medical team will tell you who to contact if you develop any side effects.
Staying close to the hospital makes it easy for you to access treatment quickly if you develop any side effects. You will also be seen frequently in the clinic during this time.
You will usually have a PET/CT scan to assess your response to treatment 1 month, 3 months and 6 months after having your CAR T cells.
You have regular follow-up appointments after you go home with your local haematology team. Your appointments get less frequent as time goes on.
Watch our CAR-T cell therapy webinar
On 9 August we held a webinar on CAR-T cell therapy, with a guest panel of a consultant haematologist, clinical nurse specialist and an individual with lived experience of this type of treatment.
