Another CAR T-cell therapy approved in Scotland
Published on: 7 October 2019Axicabtagene ciloleucel (Yescarta®) is now available on the NHS in Scotland.
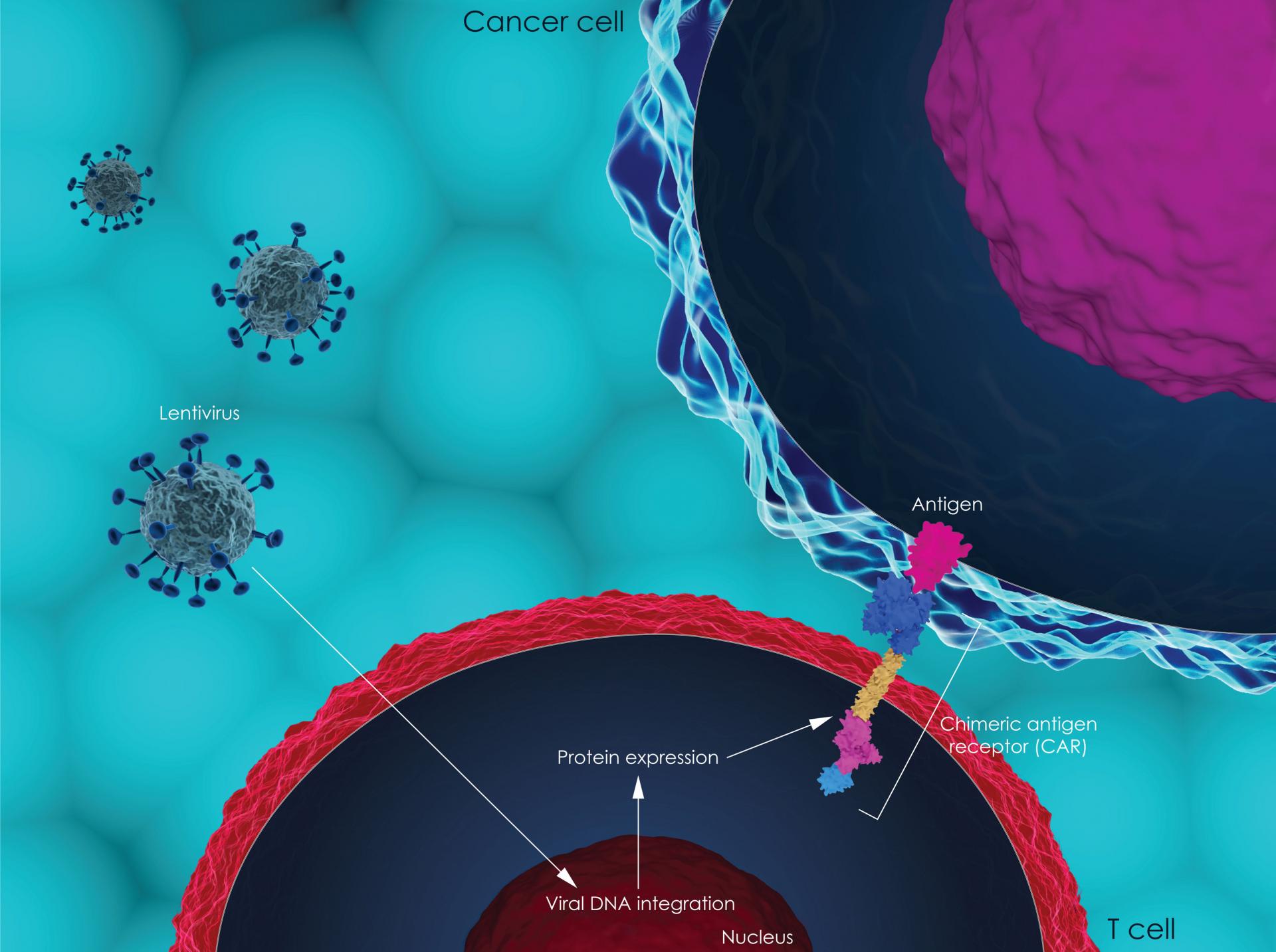
The Scottish Medicines Consortium (SMC) has today (7th October) announced that the CAR T-cell therapy axicabtagene ciloleucel (Yescarta®) will be available on the NHS in Scotland for adults with diffuse large B-cell lymphoma (DLBCL) or primary mediastinal large B cell lymphoma (PMBL) that has not responded (refractory) or has come back (relapsed) after at least two previous courses of treatment.
We are delighted at this news, which reverses the SMC’s earlier decision not to approve the therapy for use on the NHS in Scotland. Lymphoma Action was among several organisations who responded to the SMC, advocating for axicabtagene ciloleucel to be available on the NHS throughout the UK. The SMC’s reconsideration takes account of a Patient Access Scheme that improves the cost-effectiveness of axicabtagene ciloleucel.
This is further good news for people affected by lymphoma as it means there is wider access to CAR T-cell therapy in Scotland. It is particularly significant for people affected by primary mediastinal B-cell lymphoma, for whom CAR T-cell therapy would not otherwise be available in Scotland.
Although today’s decision is excellent news, there are still many challenges limiting access to CAR T-cell therapy. In the UK, CAR T-cell therapy is currently only available at a handful of approved treatment centres with the facilities and staff to administer it safely. So far all eligible patients referred for CAR T-cell therapy in the UK have had access to treatment, but we will continue to monitor the situation – especially if the number of patients who are eligible increases as we anticipate.
Lymphoma Action is collaborating with treatment centres, research groups and other patient organisations to try to improve access to CAR T-cell therapy in the UK.
A very limited number of people may be able to access CAR T-cell therapy through a clinical trial. To find out more about clinical trials for lymphoma, or to search for a trial that might be suitable for you, visit Lymphoma TrialsLink.
7 October 2019
