More good news around CAR T-cell therapy
Published on: 16 November 2018NICE recommending Kymriah for young people with B-cell acute lymphoblastic leukaemia (ALL)
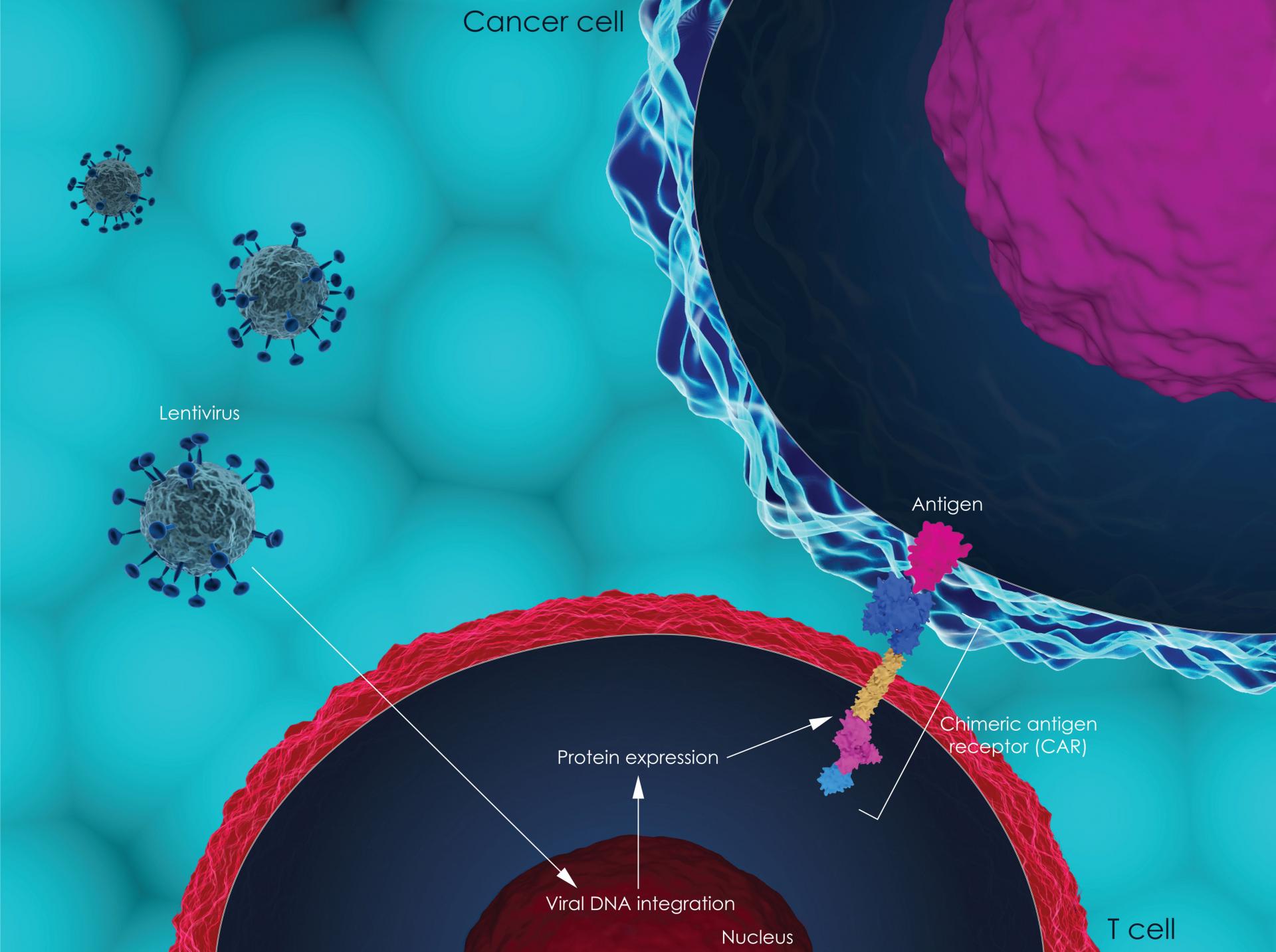
Today, NICE has announced that it is recommending tisagenlecleucel (Kymriah), a new CAR T-cell therapy, for people under 25 with B-cell acute lymphoblastic leukaemia (ALL) that has not responded to treatment (refractory) or has come back (relapsed) after a stem cell transplant. It will be available through the Cancer Drugs Fund.
This is excellent news for people with B-cell ALL but the wait is not over for people with lymphoma. NICE is currently evaluating Kymriah for people with relapsed or refractory diffuse large B-cell lymphoma (DLBCL). In its draft guidance issued in September, NICE did not recommend Kymriah for DLBCL, although it acknowledged that it can have significant clinical benefits and welcomed further discussions on its cost-effectiveness. Lymphoma Action was among several patient representative groups and professional bodies to respond to the draft guidance and we very much hope NICE will reconsider its decision.
The final NICE guidance on the use of Kymriah in the use of DLBCL is due to be published in January 2019.
November 2018
