CAR T-cell therapy not approved in Scotland
Published on: 11 March 2019Tisagenlecleucel (Kymriah®) not available for use on the NHS in Scotland
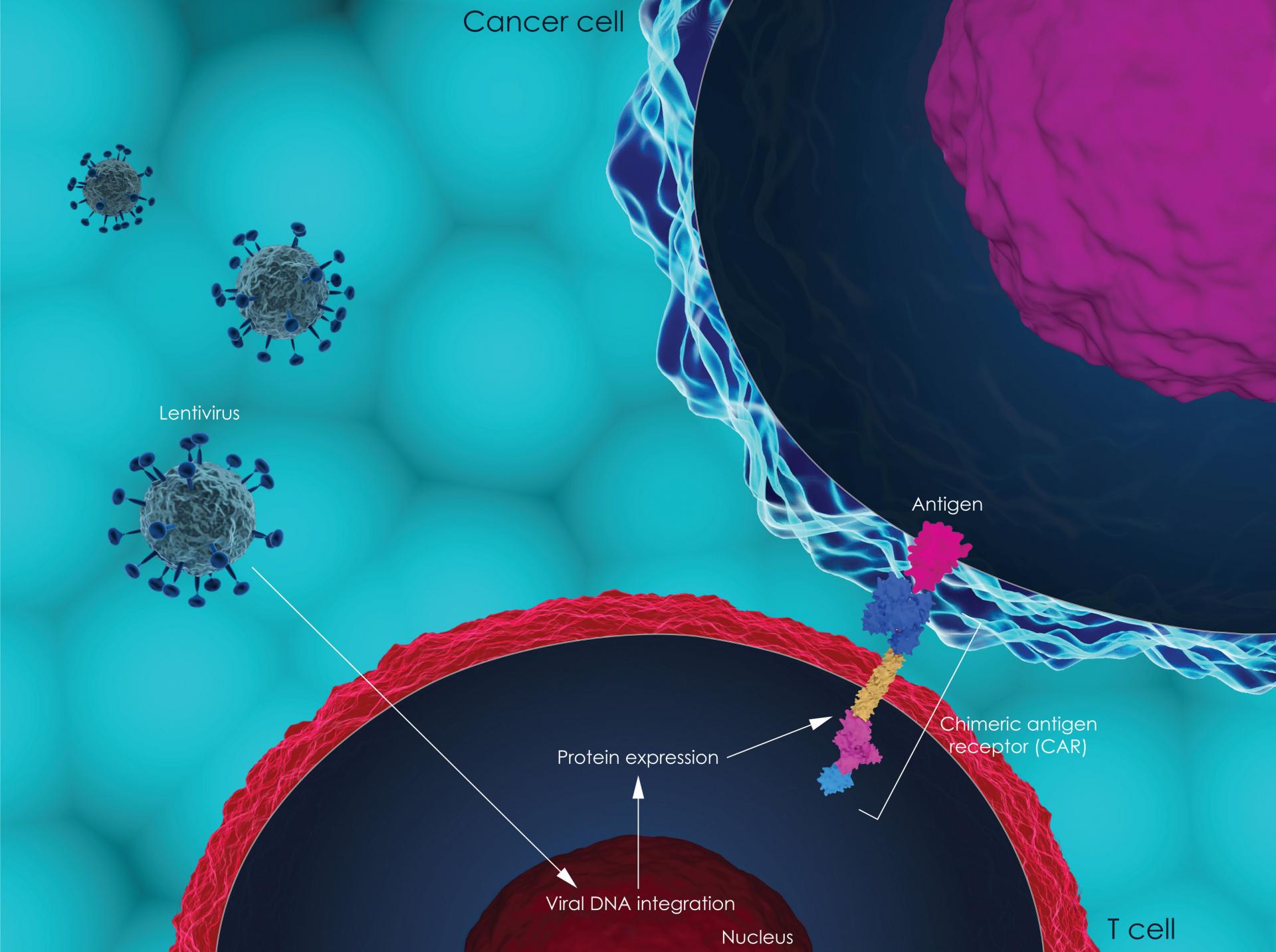
The Scottish Medicines Consortium (SMC) has today confirmed that tisagenlecleucel (Kymriah®), a CAR T-cell therapy, will not be available on the NHS in Scotland for the treatment of adults with diffuse large B-cell lymphoma (DLBCL) that has not responded (refractory) or has come back (relapsed) after two or more previous treatments.
CAR T-cell therapy is a very intensive treatment but it has shown benefits in people who otherwise have very limited treatment options. However the SMC believes there is currently not enough evidence to be certain that the health benefits of tisagenlecleucel for people with relapsed or refractory DLBCL outweigh its costs.
This follows the SMC’s decision last month not to approve axicabtagene ciloleucel (Yescarta®), another CAR T-cell therapy, for relapsed or refractory DLBCL or primary mediastinal large B-cell lymphoma.
Both these CAR T-cell therapies are available on the NHS in England and Wales to treat some people with relapsed or refractory DLBCL. It is disappointing that the decision not to approve this therapy further exacerbates inequality of access to these potentially life-saving treatments.
Novartis, the company that manufactures Kymriah®, is in ongoing communication with the SMC. In the meantime, consultants may be able to apply for special permission to use CAR T-cell therapy for people for whom it is the most appropriate treatment option through the Scottish Government’s Peer Approved Clinical System (PACS). This considers applications on a case-by-case basis.
11 March 2019
