In the news: CAR-T cell technology
Published on: 29 August 2018Our response to the NICE draft decision to reject CAR-T cell technology on the NHS.
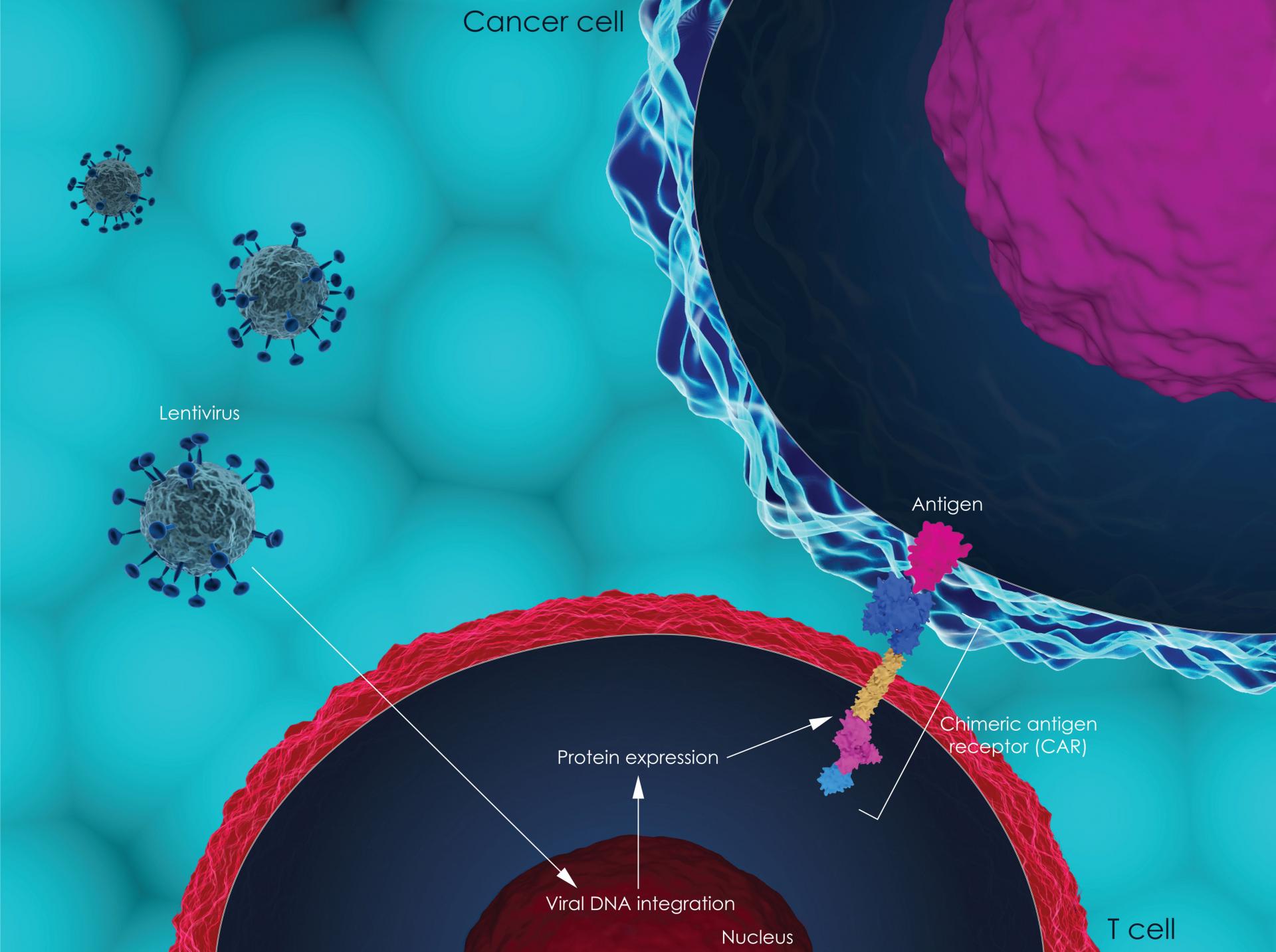
On 28 August, the National Institute for Health and Care Excellence (NICE) published draft guidance stating that they could not recommend the breakthrough CAR-T cell technology axicabtagene ciloleucel (Yescarta ®) for treating people with diffuse large B-cell lymphoma who have already received other treatments. This treatment has recently been recommended for use in Europe but this decision would mean that it would not be available on the NHS.
CAR-T cell technology is an innovative new type of treatment that has shown impressive results in some people with extremely limited treatment options, but it is very expensive and NICE considered that there was not enough evidence comparing it with current treatments.
It is important to note that NICE are only part-way through the decision-making process. There is still opportunity for the decision to be revised before the final recommendation is made. The first committee meeting allowed the committee to review the current evidence and to raise questions and concerns, which may be addressed for the next committee meeting. Lymphoma Action is preparing a response to be considered as part of the next committee meeting in late September.
Chief executive Ropinder Gill commented: 'It is disappointing that this technology has not been recommended at this stage given that its potential effectiveness is acknowledged by NICE. We hope that the pharmaceutical company will find ways to make this treatment option more affordable so that it does become a viable option for the NHS. As a charity, Lymphoma Action will continue to represent the best interests of people affected by lymphoma, whilst also providing the support that they need.'
We hope that the pharmaceutical company will find ways to make this treatment option more affordable so that it does become a viable option for the NHS
Image used under license from Shutterstock.com
